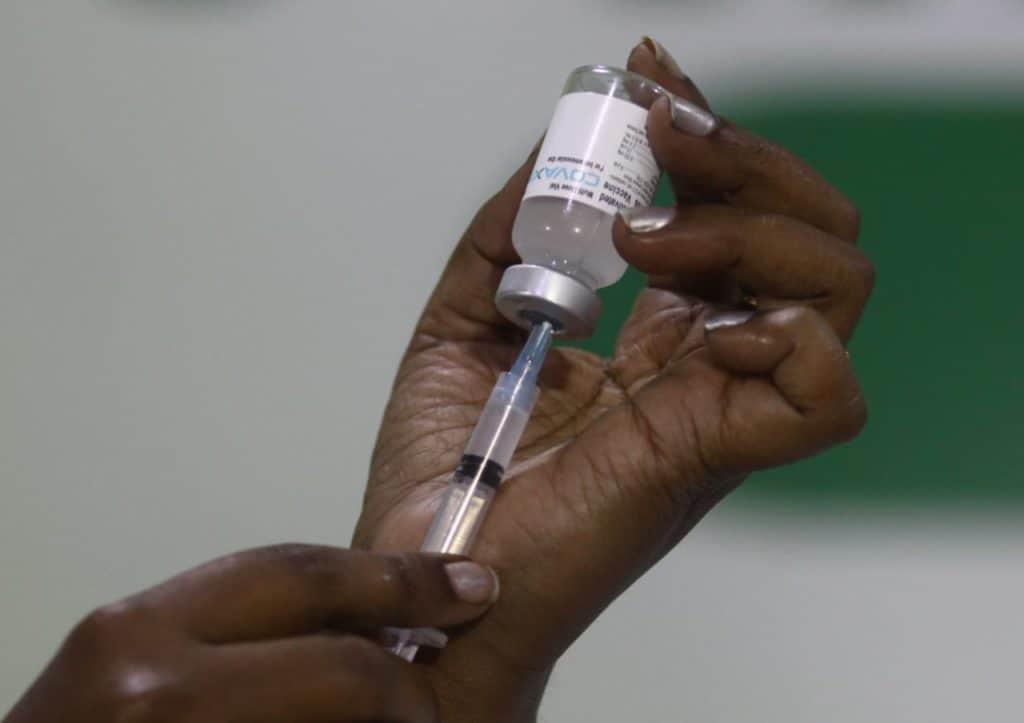
R Murugan (name changed), a nurse at the Royapettah Government Hospital, got his first dose of Covaxin at the drive held in the institution on January 16th. As a frontline worker, he was eligible to be vaccinated in the very first phase of the drive in Chennai. “Since I tend to COVID patients, it is important for me to take the vaccine and ensure that I do not pass on the viral infection to my family,” he adds.
Murugan is one of the 2,11,484 persons who got vaccinated in Tamil Nadu as of February 11th, according to data released by the state health department. The state received 5.36 lakh doses of the Oxford-AstraZeneca Covishield vaccine from the Centre, of which Chennai received 1,18,000 doses in the first phase.
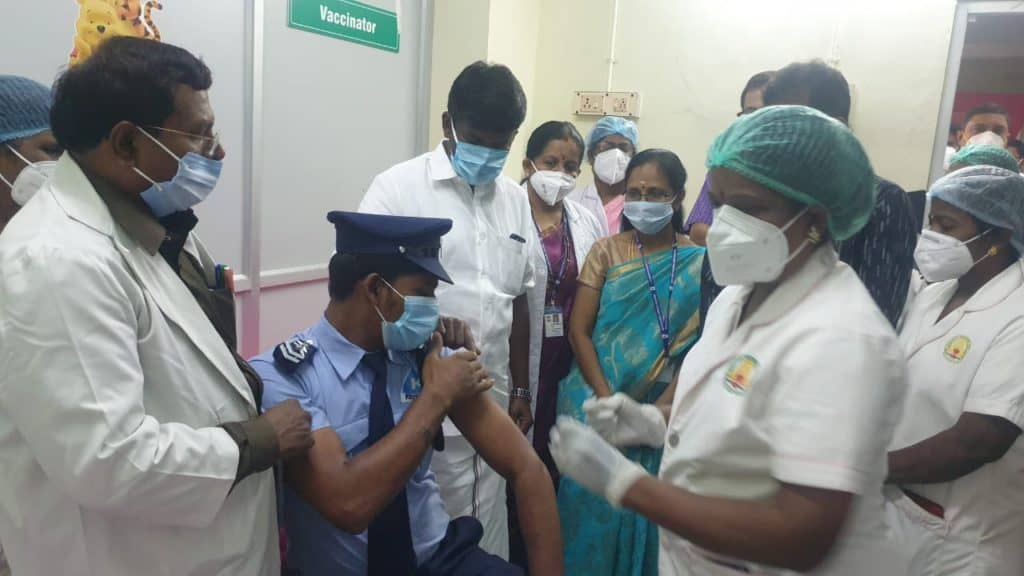
The distribution network is comprised of 359 cold chain storage points that received the 1,18,000 doses to be shared between the Greater Chennai Corporation, Poonamallee, Tiruvallur, Chengalpattu and Kancheepuram. The GCC received 63,700 doses of Covishield for distribution. Additionally, the city also received 20,000 doses of Covaxin, manufactured by Bharat Biotech, from the Central government.
The delivery of the vaccine is being carried out in four phases beginning with healthcare workers and frontline workers in the first two phases. In the latest development, the state government has announced that Public representatives, including media persons, in Tamil Nadu will be eligible for vaccination by the end of February. The general public will be vaccinated in the last phase of the drive.
Read more: All that we know about the COVID shots from the health ministry
At the vaccine site
The hospitals which are the designated vaccine centres have laid out a procedure for vaccination. Registered vaccine recipients are required to provide their photo identity proof and Aadhaar card at the registration desk and sign the consent form if the recipient gets a Covaxin shot.
“I was informed well in advance that I’d be getting the Covaxin shot and the doctors told me that the third trial is still incomplete. But since the government has cleared the vaccine, I saw no harm in taking the jab,” Murugan shares.
Once their documents are verified, recipients are asked about pre-existing allergy towards any particular medicine, after which, they are vaccinated and observed for 30 minutes to see if they exhibit any adverse reactions or anaphylaxis (also called severe allergic reactions). Every vaccination room will have an anaphylaxis kit that contains adrenaline doses.
Read more: Interview: When and how will Chennai get the COVID vaccine?
What happens after vaccination?
If a vaccine recipient does not experience any anaphylactic reactions, she is safe for discharge.
“I did not exhibit any symptoms and got back to duty after the observation period. It has been close to three weeks since I got the first shot and I had mild pain where I received the shot. Otherwise, I am doing fine,” Murugan says.
Conversations with several vaccine recipients however indicate that there is no post-vaccination follow-up from the institution, nor is there a helpline of any kind. “We have been asked to take Paracetamol tablets in case of fever or seek immediate help from the vaccine desk at Royapettah hospital, where I work, if there are any adverse symptoms,” Murugan adds.
How much do we know, really?
Being a healthcare worker, Murugan got his jab without any trouble. Since he works in a hospital, he is also reasonably well-informed and it is easy for him to seek treatment if he develops complications.
On the other side of the spectrum is R Selvi, a domestic worker. Selvi is scheduled to receive her first dose of the vaccine in the fourth phase. Largely disconnected from the developments, she is still completely unaware of the details of how and when she has to register, how the vaccine works or the controversies around it.
While the Centre mulls over the option of allowing senior citizens and people with comorbidities to self-register on Co-WIN application, (the official app rolled out for vaccine registration), to facilitate faster and easier enrolment, that is not enough cause for cheer. R Vasu is a 62-year-old carpenter in Velachery, who does not have a smartphone.
“I gather bits and pieces of news about the vaccine whenever I go out for work. The vaccine is definitely needed to protect elder people and people with comorbidities,” he says. However, when it comes to registering for a shot, Vasu finds himself in a spot as he does not own a smartphone, nor does he have enough information about what an alternative mode of registration may be.
Selvi and Vasu are not the odd ones out. Vanessa Peter, a policy researcher who works extensively with the urban poor, says that access to information is a definite challenge. “We need to change the way we look at people and devise sustainable communication strategies that will also reach the poor and vulnerable sections of society,” she adds.
Rani M from the Perumbakkam slum resettlement colony is hopeful of getting her shot, but says, “When the spread of COVID was at its peak, there were some campaigns and deployment of community volunteers by the civic body to spread awareness. But there has not been much effort ever since the vaccination camps begun. Many in the settlement do not know even the bare minimum about the importance or procedure of vaccination.”
The gap in communication cuts across sections. Greater Chennai Corporation (GCC) Commissioner G Prakash informed us in an interview that healthcare workers will be vaccinated within a month from the date of vaccine drive launch. But do we know when and how the general public will be vaccinated? Do we have a clear roadmap yet? Not really.
Even as the first phase of vaccination is underway, the civic body has not initiated any attempts to share more information on either the schedule or the process of vaccination with broader sections of the citizenry, including the marginalised.
A civic body official stated that they are awaiting guidelines from the central ministry to get started with planning strategies for the third and fourth phase. “Right now our focus is on vaccinating frontline and healthcare workers,” the official added.
Experts, however, say that expecting common citizens to be aware and well-acquainted with the larger issues around vaccine delivery and safety may be a tall ask. “Bridging that knowledge gap within a reasonable time frame may be unfeasible. Even well-experienced doctors are finding it difficult to stay abreast of the vaccine studies, as they are overwhelmed with patients already. Hence, we cannot expect all citizens to know or understand what goes behind vaccine approval and the ongoing debate about the trials here,” says Dr Vijay Gopichandran, a public health expert based in the city.
Read more: Karnataka’s vaccination drive against COVID-19 kicks off on Jan 16
Simple, digestible and bite-sized pieces of information on basic facts should, however, be disseminated to citizens, something which has not been initiated so far. “Although we are late, now is the right time to start as we are looking at a massive roll-out of the vaccine. It can be done through television commercials, messages, etc,” he adds.
Are we ready for mass vaccination?
While the country has been running the pulse polio campaign for several years now, Dr Vijay states that India is inexperienced in adult vaccination programmes.
“We are not equipped or ready to monitor adverse events following immunization or manage complications at the community level. The focus should be on community engagement and empowering communities to monitor their own neighbours,” he says.
Three weeks into the vaccine drive, Dr Vijay says the time is ripe to take proactive measures to launch an awareness campaign and train community volunteers and representatives to identify the intensity of side-effects after vaccination.
The common symptoms are mild fever, pain in the vaccinated area, all of which can be handled well with trained community volunteers and representatives. “They can be engaged in early intervention procedures. In case of adverse events such as neurological problems, the volunteers should be trained to identify the complexities and establish proper reference of a helpline efficiently,” he adds.
Also Read: